Although precise data are not available, the vast majority of beef sold in the United States today is from animals raised using conventional production practices. Some research shows that about 99 percent of beef in major retail supermarkets is from conventionally raised grain-fed cattle.
Conventional beef (also called grain-fed or grain-finished) is from cattle that spend most of their lives grazing on grass in pastures but are ‘finished’ for the last 120-200 days in a feeding operation where they receive a scientifically formulated diet of grain, roughage and nutrient supplements. Conventionally raised cattle, whether on pasture on in a feeding operation, may be given antimicrobials to prevent diseases and to treat disease if they become ill. These cattle also may be given small amounts of hormones to replace those their bodies do not produce because they have become steers (non-intact male cattle). This helps them grow more efficiently and reduces methane produced by enteric fermentation.
But consumers today have options when it comes to their food and how it’s raised. For those consumers who have shown a preference, beef producers have responded by changing production methods to provide additional choices — natural, grass-finished and organic beef. It is important to understand, however, the true differences among these production methods, and the beef they generate.
Production Differences
When it comes to safety and nutrition, there are no significant differences in the beef from animals raised in these different ways. Beef from conventionally raised cattle is basically the same as beef from animals raised in grass-finished (also called grass-fed), natural or organic operations. The difference is in the ways the animals are raised, as well as the way the beef is marketed to consumers.
The U.S. Department of Agriculture (USDA) has a specific definition for natural and specific requirements for organic beef. Based on the government’s criteria, nearly all fresh and frozen beef found in the supermarket is “natural,” meaning minimally processed and free of additives such as preservatives, artificial flavors and colors. However, the term has taken on additional meaning for some beef marketers and consumers.
For example, some marketers may use the term natural beef in reference to beef raised without use of antimicrobials and with no additional hormones. Others may use the same term to describe grass-fed or grass-finished (grass-finished cattle usually spend their entire lives on pasture).
Beef labeled “certified organic” is from animals whose production must meet a set of USDA standards. These include not being allowed to receive any antibiotics or growth promotants, and being fed only organic grass or grain. Certain vitamin and mineral supplements are permitted, as are vaccines to keep the livestock healthy. Both naturally raised and organically raised cattle may be finished in feeding operations but, in the case of organic, they must have access to pasture and be fed only organic feedstuffs.
According to the Federation of Animal Science Societies (FASS, 2003), the USDA’s National Organic Program (NOP) for meat, milk and eggs is a marketing program, not a food safety or food healthfulness program. In addition, USDA NOP regulations do not address the nutritional content of foods, food safety or animal wellbeing.
Marketers of natural, grass-finished and certified organic beef usually identify the practices that are,
or are not, employed in their production on product labels. These production practices do not make the beef safer or more nutritious.
Food Safety
It is highly unlikely that microbiological quality would be different among conventional, natural, grass-finished and organic beef processed in state or federally inspected plants (Smith et al., 2005). In fact, the entire industry has made great strides in reducing the incidence of E. coli in ground beef samples. The Food Safety and Inspection Service (FSIS) found that the percentage of E. coli O157:H7-positive ground beef samples collected in 2004 fell by 43.3 percent when compared to 2003 and declined 80 percent between 2000 and 2004. Data from 2005 and 2006 show E. coli levels in ground beef remain low, with just 0.17 percent of samples testing positive in each of the previous two years. Through the fall of 2007, the percent of samples testing positive increased to 0.23 percent. While still a small percentage of total product, it represented a 35 percent increase over 2006.
Research has not found Bovine Spongiform Encephalopathy (BSE) infectivity in beef muscle or milk. In addition, the safeguards in place protecting both animal and human health from this disease apply to all types of beef production systems. The 1997 feed ban, for example, was implemented by the Food and Drug Administration (FDA) to prevent the spread of BSE by banning the use of feed ingredients that can carry the BSE agent. These feed regulations protect all U.S. cattle equally, regardless of whether they are raised in a conventional, natural, grass finished or organic production system.
Antimicrobials
Like humans or pets, when calves or cattle become sick with a bacterial infection, they are given therapeutic doses of antimicrobials. Medicated feed containing small amounts of antimicrobials also sometimes is used to prevent diseases such as bacterial scours, liver abscesses and shipping fever. The practice keeps animals healthy. Healthy animals feel better and are likely to eat more, converting that additional feed into nutritious beef.
There is concern that use of antimicrobials in animals contributes to the development of antimicrobial-resistance that could affect human health. However, a significant body of research shows this is not the case.
Research by LeJeune and Christie (2004) suggests there is no difference between conventional beef and beef from animals raised without antibiotics for certain antibiotic-resistant organisms or multidrug resistant pathogens. Other researchers have also found that using certain antibiotics does not adversely affect the safety of the food supply (Hurd et al. 2004).
An Institute of Food Technologists (IFT) expert panel (Comprehensive Reviews in Food Science and Food Safety, 2006) concluded that focusing on antibiotic-resistant strains in food animals specifically would have far less impact on foodborne illnesses than applying interventions to control foodborne pathogens in general.
Another panel (Phillips et al., 2004) concluded that “(w)hat has not happened in 50 years of antibiotic use in animals and man, seems unlikely to happen at a rapid rate now.”
The National Cattlemen’s Beef Association (NCBA), a contractor to the Beef Checkoff, in 2004 developed “A Producer’s Guide For Judicious Use Of Antimicrobials In Cattle” for inclusion in national, state and local Beef Quality Assurance (BQA) programs. Although science does not show human health is affected by any use of antimicrobials in cattle production, producers developed these guidelines to assure that antimicrobials are used in ways that minimize any potential risk.
The National Academy of Sciences (NAS), which in 1989 concluded the low-level use of antimicrobials in animal feed to maintain health was not implicated in human illness, in 2003 concluded there should be a ban on use of antimicrobials for growth promotion in food animals if those classes of antimicrobials are also used in humans. The NCBA judicious use guidelines state that antimicrobials should be used to prevent and control disease and should not be used if the principal intent is to improve performance.
Growth Promotants
Growth promotants, most of which are naturally-occurring hormones (e.g., estrogen, testosterone, progesterone), are given in small doses to some cattle to promote growth of muscle and reduce fat deposition. The practice reduces the amount of resources required to raise cattle and helps produce the lean beef products consumers demand while keeping prices lower. In reality, growth promotants can be thought of as hormone replacement, since bulls and implanted steers (non-intact male cattle) gain at about the same rate. When used, these growth-promoting substances typically are administered in implant form as a tiny pellet placed under the skin on the back of the cattle’s ear. The active compound dissipates during the treatment period of about 80 days (depending on the specific type/kind of implant). It is important to note that all growth promoting products are subject to a stringent FDA review process which typically requires years before approval is granted. Sponsoring companies devote significant time and financial resources in research that must
prove the product’s efficacy and safety (both for cattle and humans) both prior to and following FDA approval.
Table 1. Volative residues in muscle and fat (combined) from conventional, natural and organic beef.
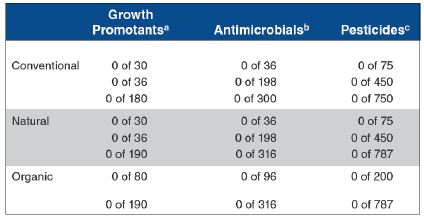
Sources: Smith et al. (1994); Usborne (1994); Smith et al. (1997) aAnabolic steroids bAntibiotics, Sulpha drugs cChlorinated hydrocarbons; organophosphates; parasiticides; pentachlorophenol
Like humans, most food animals and plants naturally produce hormones and the use of growth promotants in cattle does not create any meaningful increase in hormone levels in beef (Table 1). Two of the world’s foremost animal endocrinologists (Niswender, 2005; Veeramachaneni, 2005) believe concentrations of androgens, estrogens or progestins (natural or artificial) in beef (from cattle that were or were not implanted) are so low that there is no effect on human health associated with consuming any type of beef.
This includes any effect on puberty in children. While some research has shown girls may be reaching puberty at younger ages, there is no link between this condition and their consumption of beef from cattle given growth promotants.
Researchers have suggested a wide range of cause for earlier puberty including findings that “estrogen mimics” (environmental chemicals that resemble estrogens) (Silberstein, 2001) or “endocrine disrupters” (which disrupt proper development and function of our bodies) (Weise, 2005) may be at fault. Other research shows strong correlations between early puberty and obesity in children.
Although marketers have a right to promote the way their beef is produced and to say their beef is from animals that have never received growth promotants, all beef contains small amounts of naturally occurring hormones and science shows the hormone levels in all types of beef are safe.
Violative Residues
Through its National Residue Program (NRP), FSIS routinely tests carcasses for residues of antimicrobials, pesticides, parasiticides and the artificial hormones used in growth promoting products.
Results of the most recent NRP (FSIS-USDA, 2005) revealed that the incidence of violative chemical residues in all kinds of beef is very low, but specifically, no concerning residues of the synthetic hormones used in growth promoting products were identified. Furthermore, a product label implying meat from “animals raised free of chemicals” is misleading, since even the air animals breathe is a natural mixture of chemicals. Water they drink also is a chemical (H2O), and foods any animal consumes will contain naturally occurring chemicals.
Three studies (Table 1) have shown that violative levels of residues of growth-promoting implants, antimicrobials and pesticides are no more likely to occur in beef from conventionally produced animals than in beef from animals produced/managed to comply with “natural” production restrictions or organic standards.
The USDA Pesticide Data Program collects and analyzes more than 100,000 food samples each year, 3,400 of which are beef and chicken. Each year, less than 1 percent of samples collected have residue levels greater than tolerances established by the Environmental Protection Agency (EPA) – and the residues are more abundant in fresh fruits and vegetables than they are in meats (Punzi, et al., 2005).
Ames (1990, 1991) said of human consumption of carcinogens in the food supply: (a) 1,500 milligrams per day are “naturally occurring carcinogens,” and (b) 0.09 milligrams per day are “pesticides and synthetic chemical residues” but (c) neither exposure represents a major risk of causing cancer in humans.
The Food and Drug Administration’s Chief of Toxicology (Scheuplein, 1990) calculated that 98.8 percent of a person’s chance of contracting cancer from the food he/she eats is due to naturally occurring toxins (produced in plants for their own protection). There are no scientific data substantiating claims that because a food is lower in pesticide residues (within normal ranges), it is “safer” in a meaningful context.
Feeding of Genetically Modified (GM) Feeds
At an American Chemical Society Symposium, 12 nationally recognized experts reported (Rosen, 2004) that foods from animals fed GM crops are both safe to eat, and organic foods are no safer, healthier or more nutritious than conventional foods.
Nutritional Content
Research in the United States, United Kingdom, Canada, France and Sweden demonstrates there are few differences between conventional, natural and organic foods in nutrient composition or nutritional quality. For example, research by Woese and others (Woese et al., 1997) found that although the data were limited, there were no major differences in nutrient composition among foods produced through organic vs. conventional methods. Results of available data comparing conventional, natural, grass-finished and organic beef do not support claims of nutrient compositional differences in amounts that would be nutritionally significant to humans.
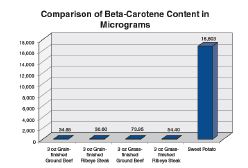
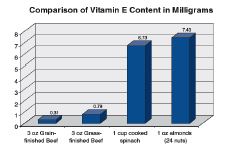
Research shows grass-finished beef has higher levels of Vitamin A, Vitamin E, omega-3 fatty acids, and conjugated linoleic acid (Duckett et al., 1993; Smith et al., 1996; French et al., 2000; Grześkiewicz et al., 2001; Poulson et al., 2004; Engle and Spears, 2004; Noci et al., 2005; Daley et al., 2005). Therefore, marketing claims that those nutrients are present in higher concentrations in grass-finished than in conventional beef are correct. However, claims that grass-finished beef is “healthier” as a result are not true, because the differences are not significant for human health (Chart 1 and 2).
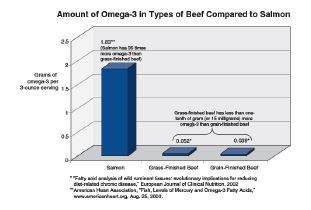
For instance, to achieve Recommended Daily Allowances and/or daily chemoprotective dietary levels of omega-3 fatty acids a person would have to eat at least 12 pounds of grass-fed beef (Rule et al., 2002; Martz et al., 2004; Guiffrida de Mendoza et al., 2005; Daley et al., 2005; Smith et al., 2005). While grassfinished beef has approximately 1/100 of a gram more omega-3 than grain-finished beef, salmon has 35 times as much omega-3 as grass-finished beef (Chart 3).
Animal Care and Handling
Consumers want assurance that their beef is from animals that are raised in a humane manner and handled with appropriate care. Livestock producers also understand that appropriate care/handling pays dividends in animal productivity and have developed a code of proper cattle care in their quality assurance programs (NCBA, 2002).
Some conventional, natural, grass-finished and organic beef programs have animal care/handling standards that go beyond those established by industry groups (Grandin, 2005). To be credible, however, such programs should be routinely audited by a third party and the beef must be from source-verified animals.
Summary
The U.S. beef industry offers products that appeal to potential customers. It accomplishes this through fresh beef identified by different USDA quality grades (Prime, Choice and Select), company brands and production methods (conventional, natural, grass-finished and organic).
The taste, texture, tenderness and other properties of products carrying these designations can vary, and marketers may capitalize on the attributes that objectively describe their products and their production methods. That’s the nature of marketing.
It is important, though, that proponents of these types of production methods not misrepresent their beef or beef from animals raised conventionally. To claim conventional beef is inferior because it contains minute additional quantities of certain chemicals (e.g., hormones or pesticides), when the amounts are insignificant and proven safe by science is not appropriate. To say that grass-finished beef is superior because it contains minute additional quantities of certain chemicals (e.g., conjugated linoleic acid or vitamin E) when it is not reasonably possible to eat enough to improve personal health, also is not appropriate.
The U.S. beef industry has a wide variety of types of beef from which consumers can choose, all of which are safe, wholesome and nutritious. Conventional, natural, grass-finished and organic beef are defined by production and marketing distinctions, not by nutritional or safety differences.
References
- Ames, B. 1991. Perspective On Risk. Food Chemical News (August Issue).
- Ames, Bruce. 1990. Carcinogens In The Food Supply. The Daily Yomiuri (Tokyo, Japan) (September 5 Issue).
- AMS-USDA. 2002. USDA-AMS, The National Organic Program. www.ams.usda.gov/nop/Consumers/brochure.html
- Daley, C.A., A. Abbott, P. Doyle, G. Nader and S. Larson. 2005. A Literature Review of the Value-Added Nutrients Found in Grass-Fed Beef Products. www.csuchico.edu/agr/grassfedbeef/health-benefits/
- Duckett, S.K., D.G. Wagner, L.D. Yates, H.G. Dolezal and S.G. May. 1993. Effects of time on feed on beef nutrient composition. J. Anim. Sci. 71:2079-2088.
- Engle, T.E. and J.W. Spears. 2004. Effect of finishing system (feedlot or pasture), high-oil maize, and copper on conjugated linoleic and other fatty acids in muscle of finishing steers.
Anim. Sci. 78:261-269. FASS. 2003. State of the Science: Organic Meat, Milk And Eggs. pp. 1-2.
- Federation of Animal Science Societies, Savoy, IL.
French, P., C. Stanton, F. Lawless, E.G. O’Riordan, F.J. Monahan, P.J. Caffrey and A.P. Moloney. 2000. Fatty acid composition, including conjugated linoleic acid, of intramuscular fat from steers offered grazed grass, grass silage or concentrate-based diets. J. Anim. Sci. 78:2849-2855.
- FSIS-USDA. 1994. Amounts Of Estrogen In Foods And People. Food Safety and Inspection Service, USDA, Washington, DC.
- FSIS-USDA. 2005. 2005 FSIS National Residue Program Data. Food Safety and Inspection Service, USDA, Washington, DC.
http://www.fsis.usda.gov/PDF/2005_Red_Book_Intro.pdf.
- Grandin, Temple. 2005. Personal communication to G.C. Smith on September 28, 2005. Dr. Grandin, Ph.D. is a Professor in the Meat Science Program of the Department of Animal Sciences,
Colorado State University, Fort Collins, CO.
- Grze śkiewicz, S., E. Bartnikowska and M.W. Obiedziński. 2001. Conjugated linoleic acid in raw round beef and beef product. Proc. Intl. Congress Meat Sci. & Technol. 47:66-67.
- Guiffrida de Mendoza, M., L. Arenas de Moreno, N. Huerta- Leidenz, S. Uzcátegui-Bracho, M.J. Beriain and G.C. Smith. 2005. Occurrence of conjugated linoleic acid in longissimus dorsi muscle of water buffalo (Bubalus bubalis) and zebu-type cattle
raised under savannah conditions. Meat Science 69:93-100.
- Hurd, H.S., S. Doores, D. Hayes, A. Mathew, J. Maurer, P. Silley, R.S. Singer and R.N. Jones. 2004. Public health consequences of macrolide use in food animals: A deterministic risk assessment. J. Food Prot. 67:980-992.
- IFT. 2006. Antimicrobial Resistance: Implications for the Food System. An Expert Report funded by the IFT Foundation. Prepublication manuscript released June 26, 2006.
- JECFA. 1987. Recommendations on ADIs and ARLs for hormonal growth promoters. Animal Pharm. No. 134, page 1 (July 24 Edition).
LeJeune, J.T. and N.P. Christie. 2004. Microbiological quality of ground beef from conventionally-reared cattle and “raised
without antibiotics” label claims. J. Food Prot. 67:1433-1437.
- Martz, F., M. Weiss, R. Kallenbach, C. Lorenzen and M. Hendrickson. 2004. Conjugated linoleic acid content of pasture finished beef and implications for human diets. Mimeographed
Report. pp. 1-9. University of Missouri, Columbia, MO
- NAS. 1987. Regulating Pesticides In Foods. National Academy of Sciences, National Research Council, National Academy Press, Washington, DC.
- NAS. 1989. Subtherapeutic Feeding Of Antibiotics. National Academy of Sciences, National Research Council, National Academy Press, Washington, DC.
- NAS. 2003. Microbial Threats To Health. National Academy of Sciences, National Research Council, National Academy Press, Washington, DC.
- NCBA. 2002. Producer Code Of Cattle Care. National Cattlemen’s Beef Association, Beef Quality Assurance Advisory Board and Cattle Health & Well-Being Committee (adopted July 2002). Centennial, CO.
- NCBA. 2004. A Producers Guide for Judicious Use of Antimicrobials in Cattle. National Cattlemen’s Beef Association, Centennial, CO. ]
- Niswender, Gordon. 2005. Personal communication; interviewed by G.C. Smith on June 28, 2005. Dr. Niswender, Ph.D., is a University Distinguished Professor at the Animal Reproduction and Biotechnology Laboratory, College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, CO.
- Noci, F., F.J. Monahan, P. French and A.P. Moloney. 2005. The fatty acid composition of muscle fat and subcutaneous adipose tissue of pasture-fed beef heifers: Influence of the duration of grazing. J. Anim. Sci. 83:1167-1178.
- Phillips, I., M. Casewell, T. Cox, B. DeGroot, C. Friis, R. Jones, C. Nightingale, R. Preston and J. Waddell. Does the use of antibiotics in food animals pose a risk to human health? A critical review of published data. 2004. J. Antimicrobial Chemotherapy, 53:28-52.
- Poulson, C.S., T.R. Dhiman, A.L. Ure, D. Cornforth and K.C. Olson. 2004. Conjugated linoleic acid content of beef from cattle fed diets containing high grain, CLA or raised on forages. Livestock Production Sciences 91:117-128.
- Punzi, J.S., M. Lamont, D. Haynes, R. L. Epstein. 2005.USDA Pesticide Data Program: Pesticide Residues on Fresh and Processed Fruits, Vegetables, Grains, Meats, Milk, and Drinking Water. Outlooks on Pesticide Management, June, 2005.
- Rosen, Joseph D. 2004. Is Organic Food Healthier Than Food? Press Release from the 228th National Meeting of the American Chemical Society (Philadelphia, PA). August 23, 2004.
- Rule, D.C., K.S. Broughton, S.M. Shellito and G. Maiorano. 2002. Comparison of muscle fatty acid profiles and cholesterol concentrations of bison, beef cattle, elk and chicken. J. Anim. Sci. 80:1202-1211.
- Scanga, J.A., J.N. Sofos, K.E. Belk, J.D. Tatum and G.C. Smith. 2004. Natural And Organic Beef. Annual Colorado State University Veterinary Conference 64:8-13.
- Scheuplein, Robert J. 1990. Food-Borne Carcinogenic Risk. Food and Drug Administration, Washington, DC.
- Silberstein, Nina. 2001. Are American Girls Reaching Puberty Sooner Than Their Grandmothers Did? Priorities for Health 13(2):42-43.
- Smith, G.C., J.N. Sofos, M.J. Aaronson, J.B. Morgan, J.D. Tatum and G.R. Schmidt. 1994. Incidence of pesticide residues and residues of chemicals specified for testing in U.S. beef by the European Community. J. Muscle Foods 5:271-284.
- Smith, G.C., J.B. Morgan, J.N. Sofos and J.D. Tatum. 1996. Supplemental Vitamin E in beef cattle diets to improve shelf-life of beef. Animal Feed Science and Technology 59:207-214.
- Smith, G.C., K.L. Heaton, J.N. Sofos, J.D. Tatum, M.J. Aaronson and R.P. Clayton. 1997. Residues of antibiotics, hormones and pesticides in conventional, natural and organic beef. J. Muscle Foods 8:157-172.
- Smith, G.C., J.D. Tatum, J.N. Sofos, K.E. Belk and J.A. Scanga. 2004. Do Organic Husbandry Practices Make Beef And Dairy Products Safer? Presented at the 228th National Meeting of the American Chemical Society (Philadelphia, PA). August 23, 2004.
- Smith, G.C., K.L. Hossner, T.E. Engle, K.E. Belk, J.A. Scanga, J.N. Sofos, T. Grandin and J.D. Tatum. 2005. Conventional, Natural, Grass-Fed And Organic Beef: Fact Sheet. pp. 1-49. Final Report to the National Cattlemen’s Beef Association, Centennial, CO. Center for Red Meat Safety, Department of Animal Sciences, Colorado State University, Fort Collins, CO.
- Veeramachaneni, D.N. Rao. 2005. Personal communication; interviewed by G.C. Smith on June 28, 2005. Dr. Veeramachaneni, Ph.D., is a Professor at the Animal Reproduction and Biotechnology Laboratory, College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, CO.
- Weise, Elizabeth. 2005b. Are Our Products Our Enemy? Chemicals In Everyday Goods Disrupt Hormones. USA Today, pp. 1D02D (August 3 Edition).
- Woese, K., D. Lange, C. Boess and K.W. Bogl. 1997. A comparison of organically and conventionally grown foods— Results of a review of the relevant literature. J. Sci. Food Agric. 74:281-293.